Coffee seems to be a part of everyday life in the United States, where more than 62% of people drink it. Intriguingly, as people are always interested in new things, drinking coffee today isn’t just about sipping a flavorful cup of coffee every morning. In fact, many individuals are passionate about learning a lot of appealing things about coffee, like how to make it in different ways or how to pair it with different foods.
Also, others are keen on the physical and chemical properties of coffee beans. Particularly, they wonder if there is any chemical reaction happening when coffee is brewed with water. Additionally, questions like “Is coffee a solution or a mixture?” is also a hot topic for many of them since both answers seem to be partly right. This article will help you figure out the detailed answers to these queries as well as what makes coffee so interesting.
Definitions Of Physical Change And Chemical Change
Before we decide whether coffee is a solution or a mixture, let’s look at what physical change and chemical changes are.
What Is Physical Change?
Physical change is when a substance changes in its shape, size, color, volume, shape, state (solid, liquid, or gas), etc., without changing its molecular composition. These changes are unstable and can be undone by simple physical methods.
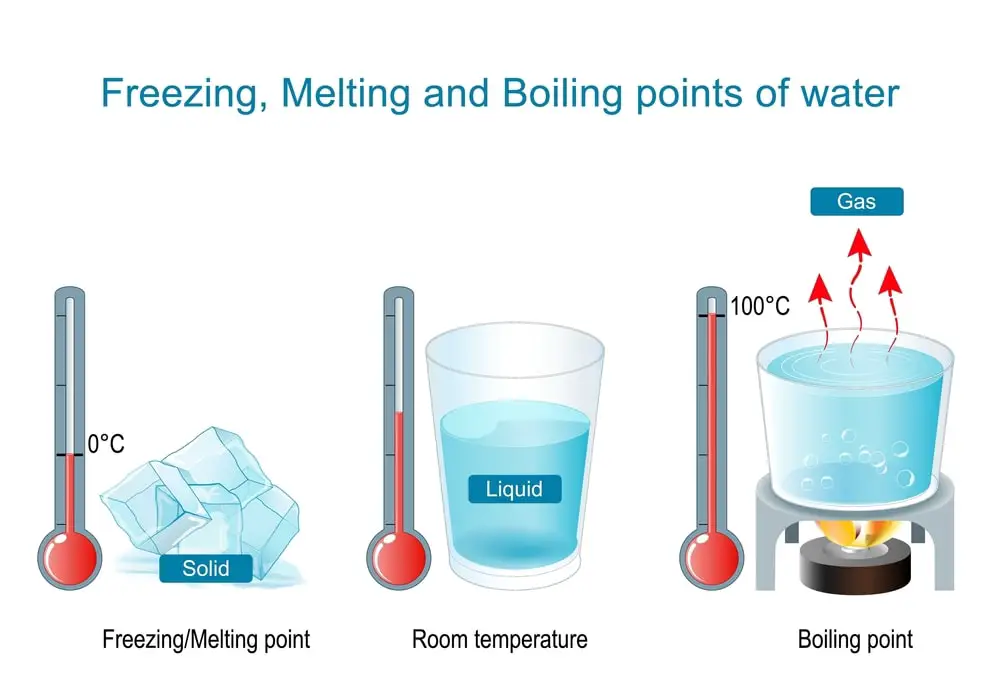
The same element or compound exists before and after the change, which means that the original qualities of the object have not altered. For example, melting wax, boiling water, dissolving sugar in water, chopping wood, crumpling paper, and so on.
What Is Chemical Change?
A chemical change is a process by which the atoms of one or more substances are rearranged or combined to make a new substance. When a substance goes through a chemical change, its chemical properties vary and it turns into another substance with a different chemical composition. Changes in energy, bubbles, smell, and temperature are some of the signs of chemical change.
A Brief Look At Solutions And Mixtures
Now that we know what physical change and chemical change mean, let’s go over the basics of mixtures and solutions.
A solution, on the other hand, is a type of mixture in which two or more parts are dissolved. This means that the solute is dissolved in a solvent.
There are many differences between these two ideas, but the most important thing to remember is that a solution can be defined and identified as a kind of mixture, but a mixture is not completely a solution sometimes.
What Is The Definition Of A Mixture In Detail?
A mixture is a thing made when two or more substances combine in a physical way, not chemically. Some typical examples are adding milk to your cereal or pouring oil into water. Obviously, even though the two substances are in the same container, they are still very different and independent to some extent. Any mixture can have a random ratio of substance A to substance B. They can also be separated with little trouble.
There are two kinds of mixtures including homogeneous mixtures and heterogeneous mixtures.
Heterogeneous Mixture
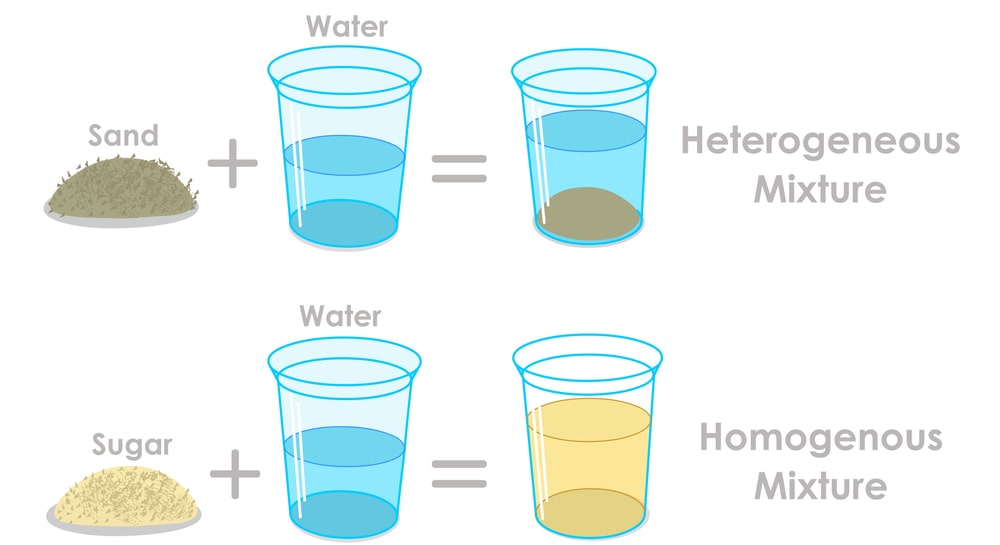
As mentioned above, heterogeneous mixtures do not have a fixed ratio of substance A to substance B. This means that they can only be a regular mixture where it is easy to separate the substances.
Homogeneous Mixture
Homogeneous mixtures tend to have a fixed ratio or amount of substance A to transparent substance B. All of this means that the substances are mixed together more closely and thoroughly.
What Is Defined As A Solution?
A solution contains two chemicals that react with each other to make a new compound. A solution is always homogeneous. Simply put, a solution has two parts, including the solute (the ingredient which will be dissolved) and the solvent (the ingredient which is responsible for dissolving the solute).
Brine, which is made when salt is mixed with water, is a basic example of a solution that is left over after this process.

In particular, coffee is a drink made from the berries of the coffee tree, which is native to countries in Africa. These berries are picked, roasted to different degrees depending on the user’s needs, and then ground into a coarse or fine powder.
When coffee and boiling water are mixed in the right amounts, they form a homogeneous mixture that is a solution. After being filtered, the water and coffee mix together completely, and we can’t tell the water from the coffee.
If Adding Milk Or Sugar To Coffee Makes It A Solution?
Coffee is a solution in and of itself, but some people wonder if sugar and milk are added, is it still a solution? In fact, coffee with milk and sugar is a homogeneous mixture (if it has been stirred well) because the ingredients are all the same all over. In other words, milk and sugar dissolve along with the coffee, leaving a mixture that is the same throughout.

Even though everything is well mixed, which means that the solutes have been properly dissolved, it is not a solution because there has been no chemical reaction that changes the properties of coffee, milk, or sugar. You can totally tell them apart by their tastes and typical traits.
Final Thought
Even though coffee is a solution in a form of a homogeneous mixture, this relatively complex definition doesn’t affect how good it tastes. Hopefully, these facts will help you learn more about this very special drink and make it more interesting to you. We’ll talk again in the next posts.
